Ammonia (data page)
In-game article clicks load inline without leaving the challenge.
This page provides supplementary chemical data on ammonia.
Structure and properties
| Molecular structure | |
|---|---|
| Point group | C3v |
| Bond length | 101.2 pm (N–H) |
| Bond angle | 106.7° (H–N–H) |
| Bond strength | 435 kJ/mol (H–NH2) |
| Crystal data | |
| Crystal structure | ? |
| Properties | |
| Dipole moment | 1.46 D |
| Dielectric constant | 22 ε0 at 239 K |
| Magnetic susceptibility | diamagnetic |
| Acidity of NH4+ (pKa) | 9.25 |
Thermodynamic properties

| Phase behavior | |
|---|---|
| Triple point | 195.4 K (−77.75 °C), 6.060 kPa |
| Critical point | 405.5 K (132.3 °C), 11.300 MPa |
| Std enthalpy change of fusion, ΔfusHo | +5.653 kJ/mol |
| Std entropy change of fusion, ΔfusSo | +28.93 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapHo | +23.35 kJ/mol at BP of −33.4 °C |
| Std entropy change of vaporization, ΔvapSo | +97.41 J/(mol·K) at BP of −33.4 °C |
| Solid properties | |
| Std enthalpy change of formation, ΔfHosolid | ? kJ/mol |
| Standard molar entropy, Sosolid | ? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfHoliquid | −80.882 ± 0.053 kJ/mol |
| Standard molar entropy, Soliquid | ? J/(mol K) |
| Heat capacity, cp | 80.80 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfHogas | −45.556 ± 0.029 kJ/mol |
| Std Gibbs free energy change of formation, ΔfGogas | −16.6 kJ/mol |
| Standard molar entropy, Sogas | 192.77 J/(mol K) |
| Heat capacity, cp | 35.06 J/(mol K) |
| Heat capacity ratio, γ at 15 °C | 1.310 |
| van der Waals' constants | a = 422.5 L2 kPa/mol2 b = 0.03707 L/mol |
Vapor–liquid equilibrium data
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15600 | 30400 | 45600 |
| T in °C | −109.1(s) | −91.9(s) | −79.2(s) | −68.4 | −45.4 | −33.6 | −18.7 | 4.7 | 25.7 | 50.1 | 78.9 | 98.3 |
Table data (above) obtained from CRC Handbook of Chemistry and Physics 44th ed. The (s) notation indicates equilibrium temperature of vapor over solid. Otherwise temperature is equilibrium of vapor over liquid.
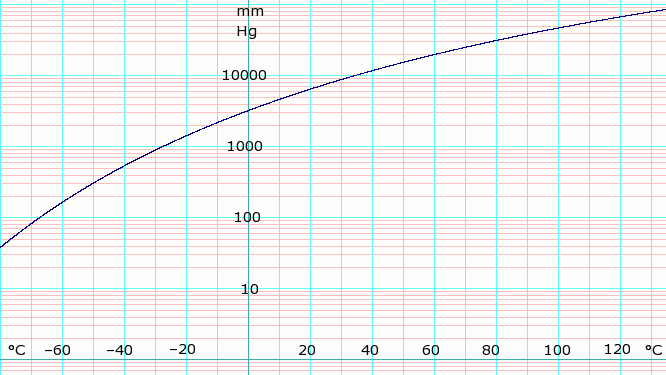
Vapor-pressure formula for ammonia:
log10P = A – B / (T − C),
where P is pressure in kPa, and T is temperature in kelvins;
A = 6.67956, B = 1002.711, C = 25.215 for T = 190 K through 333 K.
| Vapor over anhydrous ammonia | ||||
|---|---|---|---|---|
| Vapor over anhydrous ammonia Temp. Pressure ρ of liquid ρ of vapor ΔvapH −78 °C 5.90 kPa −75 °C 7.93 kPa 0.73094 g/cm3 7.8241×10−5 g/cm3 −70 °C 10.92 kPa 0.72527 g/cm3 1.1141×10−4 g/cm3 −65 °C 15.61 kPa 0.71953 g/cm3 1.5552×10−4 g/cm3 −60 °C 21.90 kPa 0.71378 g/cm3 2.1321×10−4 g/cm3 −55 °C 30.16 kPa 0.70791 g/cm3 2.8596×10−4 g/cm3 −50 °C 40.87 kPa 0.70200 g/cm3 3.8158×10−4 g/cm3 1417 J/g −45 °C 54.54 kPa 0.69604 g/cm3 4.9940×10−4 g/cm3 1404 J/g −40 °C 71.77 kPa 0.68999 g/cm3 6.4508×10−4 g/cm3 1390 J/g −35 °C 93.19 kPa 0.68385 g/cm3 8.2318×10−4 g/cm3 1375 J/g −30 °C 119.6 kPa 0.67764 g/cm3 1.0386×10−3 g/cm3 1361 J/g −25 °C 151.6 kPa 0.67137 g/cm3 1.2969×10−3 g/cm3 1345 J/g −20 °C 190.2 kPa 0.66503 g/cm3 1.6039×10−3 g/cm3 1330 J/g −15 °C 236.3 kPa 0.65854 g/cm3 1.9659×10−3 g/cm3 1314 J/g −10 °C 290.8 kPa 0.65198 g/cm3 2.3874×10−3 g/cm3 1297 J/g −5 °C 354.8 kPa 0.64533 g/cm3 2.8827×10−3 g/cm3 1280 J/g 0 °C 429.4 kPa 0.63857 g/cm3 3.4528×10−3 g/cm3 1263 J/g 5 °C 515.7 kPa 0.63167 g/cm3 4.1086×10−3 g/cm3 1245 J/g 10 °C 614.9 kPa 0.62469 g/cm3 4.8593×10−3 g/cm3 1226 J/g 15 °C 728.3 kPa 0.61755 g/cm3 5.7153×10−3 g/cm3 1207 J/g 20 °C 857.1 kPa 0.61028 g/cm3 6.6876×10−3 g/cm3 1187 J/g 25 °C 1003 kPa 0.60285 g/cm3 7.7882×10−3 g/cm3 1167 J/g 30 °C 1166 kPa 0.59524 g/cm3 9.0310×10−3 g/cm3 1146 J/g 35 °C 1350 kPa 0.58816 g/cm3 1.0431×10−2 g/cm3 1124 J/g 40 °C 1554 kPa 0.57948 g/cm3 1.2006×10−2 g/cm3 1101 J/g 45 °C 1781 kPa 0.57130 g/cm3 1.3775×10−2 g/cm3 1083 J/g 50 °C 2032 kPa 0.56287 g/cm3 1.5761×10−2 g/cm3 1052 J/g 55 °C 2310 kPa 0.55420 g/cm3 60 °C 2613 kPa 0.54523 g/cm3 2.05×10−2 g/cm3 65 °C 2947 kPa 0.53596 g/cm3 70 °C 3312 kPa 0.52632 g/cm3 2.65×10−2 g/cm3 75 °C 3711 kPa 0.51626 g/cm3 80 °C 4144 kPa 0.50571 g/cm3 3.41×10−2 g/cm3 85 °C 4614 kPa 0.49463 g/cm3 90 °C 5123 kPa 0.48290 g/cm3 4.39×10−2 g/cm3 95 °C 5672 kPa 0.47041 g/cm3 100 °C 6264 kPa 0.45693 g/cm3 5.68×10−2 g/cm3 Temp. Pressure ρ of liquid ρ of vapor ΔvapH The table above gives properties of the vapor–liquid equilibrium of anhydrous ammonia at various temperatures. The second column is vapor pressure in kPa. The third column is the density of the liquid phase. The fourth column is the density of the vapor. The fifth column is the heat of vaporization needed to convert one gram of liquid to vapor. | Freezing curve of ammonia-water system. Three eutectic points I. II. and III. are shown. Left of the I. point the frozen component is ice. Right of the III. point the frozen component is ammonia. Vapor over aqueous ammonia solution Temp. %wt NH3 Partial pressure NH3 Partial pressure H2O 0 °C 4.72 1.52 kPa 0.68 kPa 9.15 3.31 kPa 0.71 kPa 14.73 6.84 kPa 0.55 kPa 19.62 11.0 kPa 0.40 kPa 22.90 14.9 kPa 0.37 kPa 10 °C 4.16 2.20 kPa 1.21 kPa 8.26 4.96 kPa 1.17 kPa 12.32 8.56 kPa 1.01 kPa 15.88 12.68 kPa 0.93 kPa 20.54 19.89 kPa 0.83 kPa 21.83 22.64 kPa 0.73 kPa 19.9 °C 4.18 3.65 kPa 2.19 kPa 6.50 6.11 kPa 2.15 kPa 6.55 6.13 kPa 2.13 kPa 7.72 7.49 kPa 2.08 kPa 10.15 10.75 kPa 2.01 kPa 10.75 11.51 kPa 1.96 kPa 16.64 22.14 kPa 1.72 kPa 19.40 28.74 kPa 1.64 kPa 23.37 40.32 kPa 1.37 kPa 30.09 °C 3.93 5.49 kPa 4.15 kPa 7.43 11.51 kPa 3.89 kPa 9.75 16.00 kPa 3.80 kPa 12.77 23.33 kPa 3.55 kPa 17.76 38.69 kPa 3.31 kPa 17.84 38.81 kPa 3.24 kPa 21.47 53.94 kPa 2.95 kPa 40 °C 3.79 8.15 kPa 7.13 kPa 7.36 17.73 kPa 6.76 kPa 11.06 29.13 kPa 6.55 kPa 15.55 47.14 kPa 5.52 kPa 17.33 57.02 kPa 20.85 76.81 kPa 5.04 kPa 50 °C 3.29 10.54 kPa 11.95 kPa 5.90 20.17 kPa 11.61 kPa 8.91 32.88 kPa 11.07 kPa 11.57 45.56 kPa 10.75 kPa 14.15 60.18 kPa 10.27 kPa 14.94 64.94 kPa 10.03 kPa 60 °C 3.86 18.25 kPa 19.21 kPa 5.77 28.78 kPa 7.78 40.05 kPa 18.47 kPa 9.37 50.09 kPa 18.07 kPa 9.37 63.43 kPa 17.39 kPa Temp. %wt NH3 Partial Pressure NH3 Partial Pressure H2O | |||
| Temp. | Pressure | ρ of liquid | ρ of vapor | ΔvapH |
| −78 °C | 5.90 kPa | |||
| −75 °C | 7.93 kPa | 0.73094 g/cm3 | 7.8241×10−5 g/cm3 | |
| −70 °C | 10.92 kPa | 0.72527 g/cm3 | 1.1141×10−4 g/cm3 | |
| −65 °C | 15.61 kPa | 0.71953 g/cm3 | 1.5552×10−4 g/cm3 | |
| −60 °C | 21.90 kPa | 0.71378 g/cm3 | 2.1321×10−4 g/cm3 | |
| −55 °C | 30.16 kPa | 0.70791 g/cm3 | 2.8596×10−4 g/cm3 | |
| −50 °C | 40.87 kPa | 0.70200 g/cm3 | 3.8158×10−4 g/cm3 | 1417 J/g |
| −45 °C | 54.54 kPa | 0.69604 g/cm3 | 4.9940×10−4 g/cm3 | 1404 J/g |
| −40 °C | 71.77 kPa | 0.68999 g/cm3 | 6.4508×10−4 g/cm3 | 1390 J/g |
| −35 °C | 93.19 kPa | 0.68385 g/cm3 | 8.2318×10−4 g/cm3 | 1375 J/g |
| −30 °C | 119.6 kPa | 0.67764 g/cm3 | 1.0386×10−3 g/cm3 | 1361 J/g |
| −25 °C | 151.6 kPa | 0.67137 g/cm3 | 1.2969×10−3 g/cm3 | 1345 J/g |
| −20 °C | 190.2 kPa | 0.66503 g/cm3 | 1.6039×10−3 g/cm3 | 1330 J/g |
| −15 °C | 236.3 kPa | 0.65854 g/cm3 | 1.9659×10−3 g/cm3 | 1314 J/g |
| −10 °C | 290.8 kPa | 0.65198 g/cm3 | 2.3874×10−3 g/cm3 | 1297 J/g |
| −5 °C | 354.8 kPa | 0.64533 g/cm3 | 2.8827×10−3 g/cm3 | 1280 J/g |
| 0 °C | 429.4 kPa | 0.63857 g/cm3 | 3.4528×10−3 g/cm3 | 1263 J/g |
| 5 °C | 515.7 kPa | 0.63167 g/cm3 | 4.1086×10−3 g/cm3 | 1245 J/g |
| 10 °C | 614.9 kPa | 0.62469 g/cm3 | 4.8593×10−3 g/cm3 | 1226 J/g |
| 15 °C | 728.3 kPa | 0.61755 g/cm3 | 5.7153×10−3 g/cm3 | 1207 J/g |
| 20 °C | 857.1 kPa | 0.61028 g/cm3 | 6.6876×10−3 g/cm3 | 1187 J/g |
| 25 °C | 1003 kPa | 0.60285 g/cm3 | 7.7882×10−3 g/cm3 | 1167 J/g |
| 30 °C | 1166 kPa | 0.59524 g/cm3 | 9.0310×10−3 g/cm3 | 1146 J/g |
| 35 °C | 1350 kPa | 0.58816 g/cm3 | 1.0431×10−2 g/cm3 | 1124 J/g |
| 40 °C | 1554 kPa | 0.57948 g/cm3 | 1.2006×10−2 g/cm3 | 1101 J/g |
| 45 °C | 1781 kPa | 0.57130 g/cm3 | 1.3775×10−2 g/cm3 | 1083 J/g |
| 50 °C | 2032 kPa | 0.56287 g/cm3 | 1.5761×10−2 g/cm3 | 1052 J/g |
| 55 °C | 2310 kPa | 0.55420 g/cm3 | ||
| 60 °C | 2613 kPa | 0.54523 g/cm3 | 2.05×10−2 g/cm3 | |
| 65 °C | 2947 kPa | 0.53596 g/cm3 | ||
| 70 °C | 3312 kPa | 0.52632 g/cm3 | 2.65×10−2 g/cm3 | |
| 75 °C | 3711 kPa | 0.51626 g/cm3 | ||
| 80 °C | 4144 kPa | 0.50571 g/cm3 | 3.41×10−2 g/cm3 | |
| 85 °C | 4614 kPa | 0.49463 g/cm3 | ||
| 90 °C | 5123 kPa | 0.48290 g/cm3 | 4.39×10−2 g/cm3 | |
| 95 °C | 5672 kPa | 0.47041 g/cm3 | ||
| 100 °C | 6264 kPa | 0.45693 g/cm3 | 5.68×10−2 g/cm3 | |
| Temp. | Pressure | ρ of liquid | ρ of vapor | ΔvapH |
| The table above gives properties of the vapor–liquid equilibrium of anhydrous ammonia at various temperatures. The second column is vapor pressure in kPa. The third column is the density of the liquid phase. The fourth column is the density of the vapor. The fifth column is the heat of vaporization needed to convert one gram of liquid to vapor. | ||||
| Vapor over aqueous ammonia solution | ||||
| Temp. | %wt NH3 | Partial pressure NH3 | Partial pressure H2O | |
| 0 °C | 4.72 | 1.52 kPa | 0.68 kPa | |
| 9.15 | 3.31 kPa | 0.71 kPa | ||
| 14.73 | 6.84 kPa | 0.55 kPa | ||
| 19.62 | 11.0 kPa | 0.40 kPa | ||
| 22.90 | 14.9 kPa | 0.37 kPa | ||
| 10 °C | 4.16 | 2.20 kPa | 1.21 kPa | |
| 8.26 | 4.96 kPa | 1.17 kPa | ||
| 12.32 | 8.56 kPa | 1.01 kPa | ||
| 15.88 | 12.68 kPa | 0.93 kPa | ||
| 20.54 | 19.89 kPa | 0.83 kPa | ||
| 21.83 | 22.64 kPa | 0.73 kPa | ||
| 19.9 °C | 4.18 | 3.65 kPa | 2.19 kPa | |
| 6.50 | 6.11 kPa | 2.15 kPa | ||
| 6.55 | 6.13 kPa | 2.13 kPa | ||
| 7.72 | 7.49 kPa | 2.08 kPa | ||
| 10.15 | 10.75 kPa | 2.01 kPa | ||
| 10.75 | 11.51 kPa | 1.96 kPa | ||
| 16.64 | 22.14 kPa | 1.72 kPa | ||
| 19.40 | 28.74 kPa | 1.64 kPa | ||
| 23.37 | 40.32 kPa | 1.37 kPa | ||
| 30.09 °C | 3.93 | 5.49 kPa | 4.15 kPa | |
| 7.43 | 11.51 kPa | 3.89 kPa | ||
| 9.75 | 16.00 kPa | 3.80 kPa | ||
| 12.77 | 23.33 kPa | 3.55 kPa | ||
| 17.76 | 38.69 kPa | 3.31 kPa | ||
| 17.84 | 38.81 kPa | 3.24 kPa | ||
| 21.47 | 53.94 kPa | 2.95 kPa | ||
| 40 °C | 3.79 | 8.15 kPa | 7.13 kPa | |
| 7.36 | 17.73 kPa | 6.76 kPa | ||
| 11.06 | 29.13 kPa | 6.55 kPa | ||
| 15.55 | 47.14 kPa | 5.52 kPa | ||
| 17.33 | 57.02 kPa | |||
| 20.85 | 76.81 kPa | 5.04 kPa | ||
| 50 °C | 3.29 | 10.54 kPa | 11.95 kPa | |
| 5.90 | 20.17 kPa | 11.61 kPa | ||
| 8.91 | 32.88 kPa | 11.07 kPa | ||
| 11.57 | 45.56 kPa | 10.75 kPa | ||
| 14.15 | 60.18 kPa | 10.27 kPa | ||
| 14.94 | 64.94 kPa | 10.03 kPa | ||
| 60 °C | 3.86 | 18.25 kPa | 19.21 kPa | |
| 5.77 | 28.78 kPa | |||
| 7.78 | 40.05 kPa | 18.47 kPa | ||
| 9.37 | 50.09 kPa | 18.07 kPa | ||
| 9.37 | 63.43 kPa | 17.39 kPa | ||
| Temp. | %wt NH3 | Partial Pressure NH3 | Partial Pressure H2O |
Heat capacity of liquid and vapor
| Heat capacity, cp, of anhydrous ammonia gas. Uses polynomial obtained from CHERIC. |
| Heat capacity of anhydrous liquid ammonia. Uses polynomial obtained from CHERIC. |
Spectral data
| UV-Vis | |
|---|---|
| λmax | None nm |
| Extinction coefficient, ε | None |
| IR | |
| Major absorption bands | 3444, 3337, 1627, 950 cm−1 |
| NMR | |
| Proton NMR | |
| Carbon-13 NMR | None – no carbons |
| Other NMR data | |
| MS | |
| Masses of main fragments | 17 (100%) 16(80%) 15(9%) |
Regulatory data
| Regulatory data | |
|---|---|
| EINECS number | 231-635-3 (gas) 215-647-6 (soln.) |
| EU index number | 007-001-00-5 (gas) 007-001-01-2 (soln.) |
| PEL-TWA (OSHA) | 50 ppm (35 mg/m3) |
| IDLH (NIOSH) | 300 ppm |
| Flash point | 11 °C |
| Autoignition temperature | 651 °C |
| Explosive limits | 15–28% |
| RTECS # | BO0875000 |
Safety data sheet
The handling of this chemical may incur notable safety precautions... It is highly recommend that you seek the Safety Data Sheet (SDS) for this chemical from a reliable source and follow its directions.
- (Ammonia Solution)
- Linstrom, Peter J.; Mallard, William G. (eds.); , NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD)
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.
External links
- for ammonia
- (from NIST)