Please find below supplementary chemical data about dichloromethane .
MSDS sheets The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS ) for this chemical from a reliable source and follow its directions.
Structure and properties Thermodynamic properties Phase behavior Triple point ? K (? °C), ? Pa Critical point 510 K (237 °C), 6100 kPa Std enthalpy change of fusion , ΔfusH o+6.160 kJ/mol Std entropy change of fusion , ΔfusS o? J/(mol·K) Std enthalpy change of vaporization , ΔvapH o28.6 kJ/mol Std entropy change of vaporization , ΔvapS o91.43 J/(mol·K) Solid properties Std enthalpy change of formation , ΔfH osolid? kJ/mol Standard molar entropy , S osolid? J/(mol K) Heat capacity , cp ? J/(mol K) Liquid properties Std enthalpy change of formation , ΔfH oliquid−124.3 kJ/mol Standard molar entropy , S oliquid174.5 J/(mol K) Heat capacity , cp 102.3 J/(mol K) Gas properties Std enthalpy change of formation , ΔfH ogas−95.52 kJ/mol Standard molar entropy , S ogas270.28 J/(mol K) Heat capacity , cp ? J/(mol K) van der Waals' constants a = 1244 L2 kPa/mol2 b = 0.08689 liter per mole
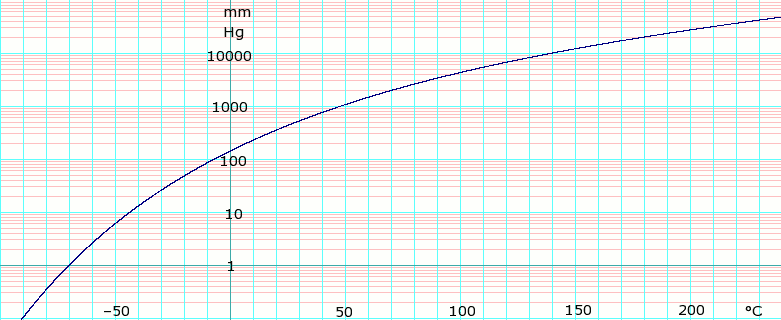
Vapor pressure of liquid P in mm Hg 1 10 40 100 400 760 T in °C –70.0 –43.3 –22.3 –6.3 24.1 40.7
Table data obtained from CRC Handbook of Chemistry and Physics 47th ed.
log10 of Dichloromethane vapor pressure. Uses formula: log e P m m H g = {\displaystyle \scriptstyle \log _{e}P_{mmHg}=}log e ( 760 101.325 ) − 10.08632 log e ( T + 273.15 ) − 6030.610 T + 273.15 + 80.87786 + 9.812512 × 10 − 6 ( T + 273.15 ) 2 {\displaystyle \scriptstyle \log _{e}({\frac {760}{101.325}})-10.08632\log _{e}(T+273.15)-{\frac {6030.610}{T+273.15}}+80.87786+9.812512\times 10^{-6}(T+273.15)^{2}} obtained from CHERICSpectral data UV-Vis Spectrum ? Lambda-max ? nm Log Ε ? IR Spectrum Major absorption bands ? cm−1 NMR Proton NMR δ CDCl3 5.30 (s, 2H) Carbon-13 NMR δ CDCl3 53.5 Other NMR data ? MS Masses of main fragments ?
Structure and properties data Except where noted otherwise, data relate to Standard temperature and pressure . Reliability of data general note. G. W. C. Kaye and T. H. Laby ,