Nonmetal
In-game article clicks load inline without leaving the challenge.
| A periodic table extract highlighting nonmetals |
| 17always/usually considered nonmetals |
| 6metalloids, sometimes considered nonmetals |
| 3status as nonmetal or metal unconfirmed |
In the context of the periodic table, a nonmetal is a chemical element that mostly lacks distinctive metallic properties. They range from colorless gases like hydrogen to shiny crystals like iodine. Physically, they are usually lighter (less dense) than elements that form metals and are often poor conductors of heat and electricity. Chemically, nonmetals have relatively high electronegativity or usually attract electrons in a chemical bond with another element, and their oxides tend to be acidic.
Seventeen elements are widely recognized as nonmetals. Additionally, some or all of six borderline elements (metalloids) are sometimes counted as nonmetals.
The two lightest nonmetals, hydrogen and helium, together account for about 98% of the mass of the observable universe. Five nonmetallic elements—hydrogen, carbon, nitrogen, oxygen, and silicon—form the bulk of Earth's atmosphere, biosphere, crust and oceans, although metallic elements are believed to be slightly more than half of the overall composition of the Earth.
Chemical compounds and alloys involving multiple elements including nonmetals are widespread. Industrial uses of nonmetals as the dominant component include in electronics, combustion, lubrication and machining.
Most nonmetallic elements were identified in the 18th and 19th centuries. While a distinction between metals and other minerals had existed since antiquity, a classification of chemical elements as metallic or nonmetallic emerged only in the late 18th century. Since then about twenty properties have been suggested as criteria for distinguishing nonmetals from metals. In contemporary research usage it is common to use a distinction between metal and not-a-metal based upon the electronic structure of the solids; the elements carbon, arsenic and antimony are then semimetals, a subclass of metals. The rest of the nonmetallic elements are insulators, some of which such as silicon and germanium can readily accommodate dopants that change the electrical conductivity leading to semiconducting behavior.
Definition and applicable elements
Unless otherwise noted, this article describes the stable form of an element at standard temperature and pressure (STP).

Nonmetallic chemical elements are often broadly defined as those that mostly lack properties commonly associated with metals—namely shininess, pliability, good thermal and electrical conductivity (due to their band structure), and a general capacity to form basic oxides. There is no widely accepted precise definition in terms of these properties; any list of nonmetals is open to debate and revision.
Fourteen elements are almost always recognized as nonmetals:
Three more are commonly classed as nonmetals, but some sources list them as "metalloids", a term which refers to elements intermediate between metals and nonmetals:
One or more of the six elements most commonly recognized as metalloids are sometimes instead counted as nonmetals:
About 15–20% of the 118 known elements are thus classified as nonmetals.
General properties
Physical

Nonmetals vary greatly in appearance, being colorless, colored or shiny. For the colorless nonmetals (hydrogen, nitrogen, oxygen, and the noble gases), no absorption of light happens in the visible part of the spectrum, and all visible light is transmitted. The colored nonmetals (sulfur, fluorine, chlorine, bromine) absorb some colors (wavelengths) and transmit the complementary or opposite colors. For example, chlorine's "familiar yellow-green colour ... is due to a broad region of absorption in the violet and blue regions of the spectrum". The shininess of boron, graphite (carbon), silicon, black phosphorus, germanium, arsenic, selenium, antimony, tellurium, and iodine is a result of the electrons reflecting incoming visible light.
About half of nonmetallic elements are gases under standard temperature and pressure; most of the rest are solids. Bromine, the only liquid, is usually topped by a layer of its reddish-brown fumes. The gaseous and liquid nonmetals have very low densities, melting and boiling points, and are poor conductors of heat and electricity. The solid nonmetals have low densities and low mechanical strength (being either hard and brittle, or soft and crumbly), and a wide range of electrical conductivity.
This diversity stems from variability in crystallographic structures and bonding arrangements. Covalent nonmetals existing as discrete atoms like xenon, or as small molecules, such as oxygen, sulfur, and bromine, have low melting and boiling points; many are gases at room temperature, as they are held together by weak London dispersion forces acting between their atoms or molecules, although the molecules themselves have strong covalent bonds. In contrast, nonmetals that form extended structures, such as long chains of selenium atoms, sheets of carbon atoms in graphite, or three-dimensional lattices of silicon atoms have higher melting and boiling points, and are all solids. Nonmetals closer to the left or bottom of the periodic table (and so closer to the metals) often have metallic interactions between their molecules, chains, or layers; this occurs in boron, carbon, phosphorus, arsenic, selenium, antimony, tellurium and iodine.
| Aspect | Metals | Nonmetals |
|---|---|---|
| Appearance and form | Shiny if freshly prepared or fractured; few colored; all but one solid | Shiny, colored or transparent; all but one solid or gaseous |
| Density | Often higher | Often lower |
| Plasticity | Mostly malleable and ductile | Often brittle solids |
| Electrical conductivity | Good | Poor to good |
| Electronic structure | Metal or semimetalic | Semimetal, semiconductor, or insulator |
Covalently bonded nonmetals often share only the electrons required to achieve a noble gas electron configuration. For example, nitrogen forms diatomic molecules featuring a triple bonds between each atom, both of which thereby attain the configuration of the noble gas neon. In contrast antimony has buckled layers in which each antimony atom is singly bonded with three other nearby atoms.
Good electrical conductivity occurs when there is metallic bonding, however the electrons in some nonmetals are not metallic. Good electrical and thermal conductivity associated with metallic electrons is seen in carbon (as graphite, along its planes), arsenic, and antimony. Good thermal conductivity occurs in boron, silicon, phosphorus, and germanium; such conductivity is transmitted though vibrations of the crystalline lattices (phonons of these elements. Moderate electrical conductivity is observed in the semiconductors boron, silicon, phosphorus, germanium, selenium, tellurium, and iodine.
Many of the nonmetallic elements are hard and brittle, where dislocations cannot readily move so they tend to undergo brittle fracture rather than deforming. Some do deform such as white phosphorus (soft as wax, pliable and can be cut with a knife at room temperature), plastic sulfur, and selenium which can be drawn into wires from its molten state. Graphite is a standard solid lubricant where dislocations move very easily in the basal planes.
Allotropes

Over half of the nonmetallic elements exhibit a range of less stable allotropic forms, each with distinct physical properties. For example, carbon, the most stable form of which is graphite, can manifest as diamond, buckminsterfullerene, amorphous and paracrystalline variations. Allotropes also occur for nitrogen, oxygen, phosphorus, sulfur, selenium and iodine.
Chemical
| Aspect | Metals | Nonmetals | |
|---|---|---|---|
| Reactivity | Wide range: very reactive to noble | ||
| Oxides | lower | Basic | Acidic; never basic |
| higher | Increasingly acidic | ||
| Compounds with metals | Alloys | Covalent or Ionic | |
| Ionization energy | Low to high | Moderate to very high | |
| Electronegativity | Low to high | Moderate to very high |
Nonmetals have relatively high values of electronegativity, and their oxides are usually acidic. Exceptions may occur if a nonmetal is not very electronegative, or if its oxidation state is low, or both. These non-acidic oxides of nonmetals may be amphoteric (like water, H2O) or neutral (like nitrous oxide, N2O), but never basic.
They tend to gain electrons during chemical reactions, in contrast to metallic elements which tend to donate electrons. This behavior is related to the stability of electron configurations in the noble gases, which have complete outer shells, empirically described by the duet and octet rules of thumb, more correctly explained in terms of valence bond theory.
The chemical differences between metals and nonmetals stem from variations in how strongly atoms attract and retain electrons. Across a period of the periodic table, the nuclear charge increases as more protons are added to the nucleus. However, because the number of inner electron shells remains constant, the effective nuclear charge experienced by the outermost electrons also increases, pulling them closer to the nucleus. This leads to a corresponding reduction in atomic radius, and a greater tendency of these elements to gain electrons during chemical reactions, forming negatively charged ions. Nonmetals, which occupy the right-hand side of the periodic table, exemplify this behavior.
Nonmetals typically exhibit higher ionization energies, electron affinities, and standard electrode potentials than metals. The higher these values are (including electronegativity) the more nonmetallic the element tends to be. For example, the chemically very active nonmetals fluorine, chlorine, bromine, and iodine have an average electronegativity of 3.19—a figure higher than that of any metallic element.
The number of compounds formed by nonmetals is vast. The first 10 places in a "top 20" table of elements most frequently encountered in 895,501,834 compounds, as listed in the Chemical Abstracts Service register for November 2, 2021, were occupied by nonmetals. Hydrogen, carbon, oxygen, and nitrogen collectively appeared in most (80%) of compounds. Silicon, a metalloid, ranked 11th. The highest-rated metal, with an occurrence frequency of 0.14%, was iron, in 12th place.
Complications
Adding complexity to the chemistry of the nonmetals are anomalies occurring in the first row of each periodic table block; non-uniform periodic trends; higher oxidation states; multiple bond formation; and property overlaps with metals.
First-row anomaly
| Condensed periodic table highlighting the first row of each block: s p d and f | ||||||||||
| Period | s-block | |||||||||
| 1 | H 1 | He 2 | p-block | |||||||
| 2 | Li 3 | Be 4 | B 5 | C 6 | N 7 | O 8 | F 9 | Ne 10 | ||
| 3 | Na 11 | Mg 12 | d-block | Al 13 | Si 14 | P 15 | S 16 | Cl 17 | Ar 18 | |
| 4 | K 19 | Ca 20 | Sc-Zn 21-30 | Ga 31 | Ge 32 | As 33 | Se 34 | Br 35 | Kr 36 | |
| 5 | Rb 37 | Sr 38 | f-block | Y-Cd 39-48 | In 49 | Sn 50 | Sb 51 | Te 52 | I 53 | Xe 54 |
| 6 | Cs 55 | Ba 56 | La-Yb 57-70 | Lu-Hg 71-80 | Tl 81 | Pb 82 | Bi 83 | Po 84 | At 85 | Rn 86 |
| 7 | Fr 87 | Ra 88 | Ac-No 89-102 | Lr-Cn 103-112 | Nh 113 | Fl 114 | Mc 115 | Lv 116 | Ts 117 | Og 118 |
| Group | (1) | (2) | (3-12) | (13) | (14) | (15) | (16) | (17) | (18) | |
| The first-row anomaly strength by block is s >> p > d > f. |
Starting with hydrogen, the first-row anomaly primarily arises from the electron configurations of the elements concerned. Hydrogen is notable for its diverse bonding behaviors. It most commonly forms covalent bonds, but it can also lose its single electron in an aqueous solution, leaving behind a bare proton with high polarizing power. Consequently, this proton can attach itself to the lone electron pair of an oxygen atom in a water molecule, laying the foundation for acid–base chemistry. Moreover, a hydrogen atom in a molecule can form a second, albeit weaker, bond with an atom or group of atoms in another molecule. Such bonding, "helps give snowflakes their hexagonal symmetry, binds DNA into a double helix; shapes the three-dimensional forms of proteins; and even raises water's boiling point high enough to make a decent cup of tea."
Hydrogen and helium, as well as boron through neon, have small atomic radii. The ionization energies and electronegativities among these elements are higher than the periodic trends would otherwise suggest.
While it would normally be expected, on electron configuration consistency grounds, that hydrogen and helium would be placed atop the s-block elements, the significant first-row anomaly shown by these two elements justifies alternative placements. Hydrogen is occasionally positioned above fluorine, in group 17, rather than above lithium in group 1. Helium is almost always placed above neon, in group 18, rather than above beryllium in group 2.
Secondary periodicity

An alternation in certain periodic trends, sometimes referred to as secondary periodicity, becomes evident when descending groups 13 to 15, and to a lesser extent, groups 16 and 17. Immediately after the first row of d-block metals, from scandium to zinc, the 3d electrons in the p-block elements—specifically, gallium (a metal), germanium, arsenic, selenium, and bromine—prove less effective at shielding the increasing positive nuclear charge.
The Soviet chemist Shchukarev[ru] gives two more tangible examples:
"The toxicity of some arsenic compounds, and the absence of this property in analogous compounds of phosphorus [P] and antimony [Sb]; and the ability of selenic acid [H2SeO4] to bring metallic gold [Au] into solution, and the absence of this property in sulfuric [H2SO4] and [H2TeO4] acids."
Higher oxidation states
Roman numerals such as III, V and VIII denote oxidation states
Some nonmetallic elements exhibit oxidation states that deviate from those predicted by the octet rule, which typically results in an oxidation state of –3 in group 15, –2 in group 16, –1 in group 17, and 0 in group 18. Examples include ammonia NH3, hydrogen sulfide H2S, hydrogen fluoride HF, and elemental xenon Xe. Meanwhile, the maximum possible oxidation state increases from +5 in group 15, to +8 in group 18. The +5 oxidation state is observable from period 2 onward, in compounds such as nitric acid HN(V)O3 and phosphorus pentafluoride PCl5. Higher oxidation states in later groups emerge from period 3 onwards, as seen in sulfur hexafluoride SF6, iodine heptafluoride IF7, and xenon(VIII) tetroxide XeO4. For heavier nonmetals, their larger atomic radii and lower electronegativity values enable the formation of compounds with higher oxidation numbers, supporting higher bulk coordination numbers.
Multiple bond formation
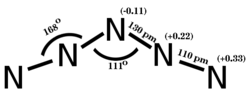
Period 2 nonmetals, particularly carbon, nitrogen, and oxygen, show a propensity to form multiple bonds. The compounds formed by these elements often exhibit unique stoichiometries and structures, as seen in the various nitrogen oxides, which are not commonly found in elements from later periods.
Property overlaps
While certain elements have traditionally been classified as nonmetals and others as metals, some overlapping of properties occurs. Writing early in the twentieth century, by which time the era of modern chemistry had been well-established (although not as yet more precise quantum chemistry) Humphrey observed that:
... these two groups, however, are not marked off perfectly sharply from each other; some nonmetals resemble metals in certain of their properties, and some metals approximate in some ways to the non-metals.

Examples of metal-like properties occurring in nonmetallic elements include:
- Silicon has an electronegativity (1.9) comparable with metals such as cobalt (1.88), copper (1.9), nickel (1.91) and silver (1.93);
- The electrical conductivity of graphite exceeds that of some metals;
- Selenium can be drawn into a wire;
- Radon is the most metallic of the noble gases and begins to show some cationic behavior, which is unusual for a nonmetal; and
- In extreme conditions, just over half of nonmetallic elements can form homopolyatomic cations.
Examples of nonmetal-like properties occurring in metals are:
- Tungsten displays some nonmetallic properties, sometimes being brittle, having a high electronegativity, and forming only anions in aqueous solution, and predominately acidic oxides.
- Gold, the "king of metals" has the highest electrode potential among metals, suggesting a preference for gaining rather than losing electrons. Gold's ionization energy is one of the highest among metals, and its electron affinity and electronegativity are high, with the latter exceeding that of some nonmetals. It forms the Au– auride anion and exhibits a tendency to bond to itself, behaviors which are unexpected for metals. In aurides (MAu, where M = Li–Cs), gold's behavior is similar to that of a halogen. The reason for this is that gold has a large enough nuclear potential that the electrons have to be considered with relativistic effects included, which changes some of the properties.
A relatively recent development involves certain compounds of heavier p-block elements, such as silicon, phosphorus, germanium, arsenic and antimony, exhibiting behaviors typically associated with transition metal complexes. This is linked to a small energy gap between their filled and empty molecular orbitals, which are the regions in a molecule where electrons reside and where they can be available for chemical reactions. In such compounds, this allows for unusual reactivity with small molecules like hydrogen (H2), ammonia (NH3), and ethylene (C2H4), a characteristic previously observed primarily in transition metal compounds. These reactions may open new avenues in catalytic applications.
Types
Nonmetal classification schemes vary widely, with some accommodating as few as two subtypes and others up to seven. For example, the periodic table in the Encyclopaedia Britannica recognizes noble gases, halogens, and other nonmetals, and splits the elements commonly recognized as metalloids between "other metals" and "other nonmetals". On the other hand, seven of twelve color categories on the Royal Society of Chemistry periodic table include nonmetals.
Starting on the right side of the periodic table, three types of nonmetals can be recognized:
The elements in a fourth set are sometimes recognized as nonmetals:
While many of the early workers attempted to classify elements none of their classifications were satisfactory. They were divided into metals and nonmetals, but some were soon found to have properties of both. These were called metalloids. This only added to the confusion by making two indistinct divisions where one existed before.
The boundaries between these types are not sharp. Carbon, phosphorus, selenium, and iodine border the metalloids and show some metallic character, as does hydrogen.
The greatest discrepancy between authors occurs in metalloid "frontier territory". Some consider metalloids distinct from both metals and nonmetals, while others classify them as nonmetals. Some categorize certain metalloids as metals (e.g., arsenic and antimony due to their similarities to heavy metals). Metalloids resemble the elements universally considered "nonmetals" in having relatively low densities, high electronegativity, and similar chemical behavior.
Noble gases

Six nonmetals are classified as noble gases: helium, neon, argon, krypton, xenon, and the radioactive radon. In conventional periodic tables they occupy the rightmost column. They are called noble gases due to their exceptionally low chemical reactivity.
These elements exhibit similar properties, being colorlessness, odorless, and nonflammable. Due to their closed outer electron shells, noble gases possess weak interatomic forces of attraction, leading to exceptionally low melting and boiling points.
Chemically, the noble gases exhibit relatively high ionization energies, negligible or negative electron affinities, and high to very high electronegativities. The number of compounds formed by noble gases is in the hundreds and continues to expand, with most of these compounds involving the combination of oxygen or fluorine with either krypton, xenon, or radon.
Halogen nonmetals
Chemically, the halogen nonmetals have high ionization energies, electron affinities, and electronegativity values, and are relatively strong oxidizing agents. All four elements tend to form primarily ionic compounds with metals, in contrast to the remaining nonmetals (except for oxygen) which tend to form primarily covalent compounds with metals.
Unclassified nonmetals

Hydrogen behaves in some respects like a metallic element and in others like a nonmetal. Like a metallic element it can, for example, form a solvated cation in aqueous solution; it can substitute for alkali metals in compounds such as the chlorides (NaCl cf. HCl) and nitrates (KNO3 cf. HNO3), and in certain alkali metal complexes as a nonmetal. It attains this configuration by forming a covalent or ionic bond or by bonding as an ion to a lone pair of electrons.
Some or all of these nonmetals share several properties. Being generally less reactive than the halogens, most of them can occur naturally in the environment. Collectively, their physical and chemical characteristics can be described as "moderately non-metallic". When combined with metals, the unclassified nonmetals can form interstitial or refractory compounds. They also exhibit a tendency to bond to themselves, particularly in solid compounds. Additionally, diagonal periodic table relationships among these nonmetals mirror similar relationships among the metalloids.
Abundance, extraction, and uses
Abundance
| Universe | 75% *hydrogen | 23% *helium | 1% *oxygen |
| Atmosphere | 78% *nitrogen | 21% *oxygen | 0.5% *argon |
| Hydrosphere | 86% *oxygen | 11% *hydrogen | 2% *chlorine |
| Biomass | 63% *oxygen | 20% *carbon | 10% *hydrogen |
| Crust | 46% *oxygen | 27% *silicon | 8% aluminium |
| Earth | 32% iron | 30% *oxygen | 14% magnesium |
The abundance of elements in the universe results from nuclear physics processes like nucleosynthesis and radioactive decay.
The volatile noble gas nonmetal elements are less abundant in the atmosphere than expected based upon their overall abundance due to cosmic nucleosynthesis. Mechanisms to explain this difference is an important aspect of planetary science. The element Xe is unexpectedly depleted, and a possible explanation comes from theoretical models of the high pressures in the Earth's core suggesting that there may be around 1013 tons of xenon in the form of stable XeFe3 and XeNi3 intermetallic compounds.
Five nonmetals—hydrogen, carbon, nitrogen, oxygen, and silicon—form the bulk of the directly observable structure of the Earth: about 73% of the crust, 93% of the biomass, 96% of the hydrosphere, and over 99% of the atmosphere, as shown in the accompanying table. Silicon and oxygen form stable tetrahedral structures, known as silicates. Here, "the powerful bond that unites the oxygen and silicon ions is the cement that holds the Earth's crust together." However, they make up less than 50% of the total weight of the earth.
In the biomass, the relative abundance of the first four nonmetals (and phosphorus, sulfur, and selenium marginally) is attributed to a combination of relatively small atomic size, and sufficient spare electrons. These two properties enable them to bind to one another and "some other elements, to produce a molecular soup sufficient to build a self-replicating system".
Extraction
Nine of the 23 nonmetallic elements are gases, or form compounds that are gases, and are extracted from natural gas or liquid air, including hydrogen, nitrogen, oxygen, sulfur, and most of the noble gases. For example, nitrogen and oxygen are extracted from liquid air through fractional distillation and sulfur from the hydrogen sulfide in natural gas by reacting it with oxygen to yield water and sulfur. Three nonmetals are extracted from seawater; the rest of the nonmetals – and almost all metals – from mining solid ores.[citation needed]
| Group (1, 13−18) | Period | ||||||
| 13 | 14 | 15 | 16 | 1/17 | 18 | (1−6) | |
| H | He | 1 | |||||
| B | C | N | O | F | Ne | 2 | |
| Si | P | S | Cl | Ar | 3 | ||
| Ge | As | Se | Br | Kr | 4 | ||
| Sb | Te | I | Xe | 5 | |||
| Rn | 6 | ||||||
Nonmetallic elements are extracted from these sources:
3from natural gas components
hydrogen (methane), helium, and sulfur (hydrogen sulfide)
6from liquefied air
nitrogen, oxygen, neon, argon, krypton, and xenon
3from seawater brine
chlorine, bromine, and iodine
12from solid ores
boron (borates), carbon (natural graphite), silicon (silica), phosphorus (phosphates), iodine (sodium iodate), radon (uranium ore decay product), fluorine (fluorite); and germanium, arsenic, selenium, antimony, and tellurium (from their sulfides).
Uses

Nonmetallic elements are present in combination with other elements in almost everything around us, from water to plastics and within metallic alloys. There are some specific uses of the elements themselves, although these are less common; extensive details can be found in the specific pages of the relevant elements. A few examples are:
- Hydrogen can be used in fuel cells, and is being explored for a possible future low-carbon hydrogen economy.
- Carbon has many uses, ranging from decorative applications of diamond jewelry to diamond in cutting blades and graphite as a solid lubricant.
- Liquid nitrogen is extensively used as a coolant.
- Oxygen is a critical component of the air we breath. (While nitrogen is also present, it is less used from the air, mainly by certain bacteria.) Oxygen gas and liquid is also heavily used for combustion in welding and cutting torches and as a component of rocket fuels.
- Silicon is the most widely used semiconductor. While ultra-pure silicon is an insulator, by selectively adding electronic dopants it can be used as a semiconductor where the chemical potential of the electrons can be manipulated, this being exploited in a wide range of electronic devices.
- The noble gases have a range of applications, including liquid helium for cryogenic cooling, and argon to in gaseous fire suppression to -damp fires around sensitive electrical equipment where water cannot be used.
- Radon is a potentially hazardous indoor pollutant.
Taxonomical history
Background
Medieval chemical philosophers focused on metals, rarely investigating nonmetallic minerals.

Organization of elements by types
In the late 1700s, French chemist Antoine Lavoisier published the first modern list of chemical elements in his revolutionary 1789 Traité élémentaire de chimie. The 33 elements known to Lavoisier were categorized into four distinct groups, including gases, metallic substances, nonmetallic substances that form acids when oxidized, and earths (heat-resistant oxides). Lavoisier's work gained widespread recognition and was republished in twenty-three editions across six languages within its first seventeen years, significantly advancing the understanding of chemistry in Europe and America. Lavoisier's chemistry was "dualistic",: "salts" were combinations of "acid" and "base"; acids where combinations of oxygen and metals while bases where combinations of oxygen and nonmetals. This view prevailed despite increasing evidence that chemicals like chlorine and ammonia contained no oxygen, in large part due the vigious if sometimes misguided defense by the Swedish chemist Berzelius.
In 1802 the term "metalloids" was introduced for elements with the physical properties of metals but the chemical properties of non-metals. In 1811 Berzelius used the term "metalloids" to describe all nonmetallic elements, noting their ability to form negatively charged ions with oxygen in aqueous solutions. Drawing on this, in 1864 the "Manual of Metalloids" divided all elements into either metals or metalloids, with the latter group including elements now called nonmetals. Reviews of the book indicated that the term "metalloids" was still endorsed by leading authorities, but there were reservations about its appropriateness. While Berzelius' terminology gained significant acceptance, it later faced criticism from some who found it counterintuitive, misapplied, or even invalid. The idea of designating elements like arsenic as metalloids had been considered. The use of the term "metalloids" persisted in France as textbooks of chemistry appeared in the 1800s. During this period, "metals" as a chemical category were characterized by a single property, their affinity for oxygen, while "metalloids" were organized by comparison of many characteristic analogous to the approach of naturalists.
Development of types

In 1844, Alphonse Dupasquier[fr], a French doctor, pharmacist, and chemist, established a basic taxonomy of nonmetals to aid in their study. He wrote:
They will be divided into four groups or sections, as in the following: Organogens—oxygen, nitrogen, hydrogen, carbon Sulphuroids—sulfur, selenium, phosphorus Chloroides—fluorine, chlorine, bromine, iodine Boroids—boron, silicon.
Dupasquier's quartet parallels the modern nonmetal types. The organogens and sulphuroids are akin to the unclassified nonmetals. The chloroides were later called halogens. The boroids eventually evolved into the metalloids, with this classification beginning from as early as 1864. The then unknown noble gases were recognized as a distinct nonmetal group after being discovered in the late 1800s. This taxonomy was noted as a "natural classification" of the substance considering all aspects rather than an single characteristic like oxygen affinity. It was a significant departure from other contemporary classifications, since it grouped together oxygen, nitrogen, hydrogen, and carbon.
In 1828 and 1859, the French chemist Dumas classified nonmetals as (1) hydrogen; (2) fluorine to iodine; (3) oxygen to sulfur; (4) nitrogen to arsenic; and (5) carbon, boron and silicon, thereby anticipating the vertical groupings of Mendeleev's 1871 periodic table. Dumas' five classes fall into modern groups 1, 17, 16, 15, and 14 to 13 respectively.
Nonmetals as terminology
By as early as 1866, some authors began preferring the term "nonmetal" over "metalloid" to describe nonmetallic elements. In 1875, Kemshead observed that elements were categorized into two groups: non-metals (or metalloids) and metals. He noted that the term "non-metal", despite its compound nature, was more precise and had become universally accepted as the nomenclature of choice.
Structure, quantum mechanics and band structure
The early terminologies were empirical categorizations based upon observables. As the 20th century started there were significant changes in understanding. The first was due to methods, mainly x-ray crystallography, for determining how atoms are arranged in the various materials. As early as 1784 René Just Haüy discovered that every face of a crystal could be described by simple stacking patterns of blocks of the same shape and size (law of decrements). Haüy's study led to the idea that crystals are a regular three-dimensional array (a Bravais lattice) of atoms and molecules, with a single unit cell repeated indefinitely, along with other developments in the early days of physical crystallography. After Max von Laue demonstrated in 1912 that x-rays diffract, fairly quickly William Lawrence Bragg and his father William Henry Bragg were able to solve previously unknown structures. Building on this, it became clear that most of the simple elemental metals had close packed structures. With this determined the concept of dislocations originally developed by Vito Volterra in 1907 became accepted, for instance being used to explain the ductility of metals by G. I. Taylor in 1934.
The second was the advent of quantum mechanics. In 1924 Louis de Broglie in his PhD thesis Recherches sur la théorie des quanta introduced his theory of electron waves. This rapidly became part of what was called by Erwin Schrödinger undulatory mechanics, now called the Schrödinger equation, wave mechanics or more commonly in contemporary usage quantum mechanics. While it was not so easy to solve the mathematics in the early days, fairly rapidly ideas such as the chemical bond terminology of Linus Pauling as well as electronic band structure concepts were developed.

From this the concept of nonmetals as "not-a-metal" originates. The original approach to describe metals and nonmetals was a band-structure with delocalized electrons (i.e. spread out in space). A nonmetal has a gap in the energy levels of the electrons at the Fermi level. In contrast, a metal would have at least one partially occupied band at the Fermi level; in a semiconductor or insulator there are no delocalized states at the Fermi level, see for instance Ashcroft and Mermin. (A semimetal is similar to a metal, with a slightly more complex band structure.) These definitions are equivalent to stating that metals conduct electricity at absolute zero, as suggested by Nevill Francis Mott, and the equivalent definition at other temperatures is also commonly used as in textbooks such as Chemistry of the Non-Metals by Ralf Steudel and work on metal–insulator transitions.
Originally this band structure interpretation was based upon a single-electron approach with the Fermi level in the band gap as illustrated in the Figure, not including a complete picture of the many-body problem where both exchange and correlation terms matter, as well as relativistic effects such as spin-orbit coupling. For instance, the passivity of gold is typically associated with relativistic terms. A key addition by Mott and Rudolf Peierls was that these could not be ignored. For instance, nickel oxide would be a metal if a single-electron approach was used, but in fact has quite a large band gap. As of 2024 it is more common to use an approach based upon density functional theory where the many-body terms are included. Although accurate calculations remain a challenge, reasonable results are now available in many cases.
It is common to nuance the early definition of Alan Herries Wilson and Mott which was for a zero temperature. As discussed by Peter Edwards and colleagues, as well as Fumiko Yonezawa,it is important to consider the temperatures at which both metals and nonmetals are used. Yonezawa provides a general definition for both general temperatures and conditions (such as standard temperature and pressure):
When a material conducts and at the same time the temperature coefficient of the electric conductivity of that material is not positive under a certain environmental condition, the material is metallic under that environmental condition. A material which does not satisfy these requirements is not metallic under that environmental condition.
The precise meaning of semiconductor needs a little care. In terms of the temperature dependence of their conductivity they are all classified as insulators; the pure forms are intrinsic semiconductors. When they are doped their conductivity continues to increase with temperature, and can become substantial; hence the ambiguities with an empirical categorisation using conductivity described earlier. Indeed, some elements that are normally considered as insulators have been exploited as semiconductors. For instance diamond, which has the largest band gap of the elements that are solids under normal conditions, has a number of semiconductor applications.
Band structure definitions of metals and nonmetals are widely used in current research into materials, and apply both to single elements such as insulating boron as well as compounds such as strontium titanate. The characteristics associated with metals and nonmetals in early work such as their appearance and mechanical properties are now understood to be consequences of how the atoms and electrons are arranged.
Comparison of selected properties
The two tables in this section list some of the properties of five types of elements (noble gases, halogen nonmetals, unclassified nonmetals, metalloids and, for comparison, metals) based on their most stable forms at standard temperature and pressure. The dashed lines around the columns for metalloids signify that the treatment of these elements as a distinct type can vary depending on the author, or classification scheme in use.
Physical properties by element type
Physical properties are listed in loose order of ease of their determination.
| Property | Element type | ||||
|---|---|---|---|---|---|
| Metals | Metalloids | Unc. nonmetals | Halogen nonmetals | Noble gases | |
| General physical appearance | lustrous | lustrous | ◇ lustrous: carbon, phosphorus, selenium ◇ colored: sulfur ◇ colorless: hydrogen, nitrogen, oxygen | ◇ lustrous: iodine ◇ colored: fluorine, chlorine, bromine | colorless |
| Form and density | solid (Hg liquid) | solid | solid or gas | solid or gas (bromine liquid) | gas |
| often high density such as iron, lead, tungsten | low to moderately high density | low density | low density | low density | |
| some light metals including beryllium, magnesium, aluminium | all lighter than iron | hydrogen, nitrogen lighter than air | helium, neon lighter than air | ||
| Plasticity | mostly malleable and ductile | often brittle | phosphorus, sulfur, selenium, brittle | iodine brittle | not applicable |
| Electrical conductivity | good | ◇ moderate: boron, silicon, germanium, tellurium ◇ good: arsenic, antimony | ◇ poor: hydrogen, nitrogen, oxygen, sulfur ◇ moderate: phosphorus, selenium ◇ good: carbon | ◇ poor: fluorine, chlorine, bromine ◇ moderate: I | poor |
| Electronic structure | metal (beryllium, strontium, α-tin, ytterbium, bismuth are semimetals) | semimetal (arsenic, antimony) or semiconductor | ◇ semimetal: carbon ◇ semiconductor: phosphorus ◇ insulator: hydrogen, nitrogen, oxygen, sulfur | semiconductor (I) or insulator | insulator |
Chemical properties by element type
Chemical properties are listed from general characteristics to more specific details.
| Property | Element type | ||||
|---|---|---|---|---|---|
| Metals | Metalloids | Unc. nonmetals | Halogen nonmetals | Noble gases | |
| General chemical behavior | ◇ strong to weakly metallic ◇ noble metals are relatively inert | weakly nonmetallic | moderately nonmetallic | strongly nonmetallic | ◇ inert to nonmetallic ◇ radon shows some cationic behavior |
| Oxides | basic; some amphoteric or acidic | amphoteric or weakly acidic | acidic or neutral | acidic | metastable XeO3 is acidic; stable XeO4 strongly so |
| few glass formers | all glass formers | some glass formers | no glass formers reported | no glass formers reported | |
| ionic, polymeric, layer, chain, and molecular structures | polymeric in structure | ◇ mostly molecular ◇ carbon, phosphorus, sulfur, selenium have 1+ polymeric forms | ◇ mostly molecular ◇ iodine has a polymeric form, I2O5 | ◇ mostly molecular ◇ XeO2 is polymeric | |
| Compounds with metals | alloys or intermetallic compounds | tend to form alloys or intermetallic compounds | ◇ salt-like to covalent or metallic: hydrogen†, carbon, nitrogen, phosphorus, sulfur, selenium ◇ mainly ionic: oxygen | mainly ionic | simple compounds at STP not known |
| Ionization energy (kJ mol−1)‡ | low to high | moderate | moderate to high | high | high to very high |
| 376 to 1,007 | 762 to 947 | 941 to 1,402 | 1,008 to 1,681 | 1,037 to 2,372 | |
| average 643 | average 833 | average 1,152 | average 1,270 | average 1,589 | |
| Electronegativity (Pauling)‡ | low to high | moderate | moderate to high | high | high (radon) to very high |
| 0.7 to 2.54 | 1.9 to 2.18 | 2.19 to 3.44 | 2.66 to 3.98 | ca. 2.43 to 4.7 | |
| average 1.5 | average 2.05 | average 2.65 | average 3.19 | average 3.3 |
† Hydrogen can also form alloy-like hydrides ‡ The labels low, moderate, high, and very high are arbitrarily based on the value spans listed in the table
See also
- CHON (carbon, hydrogen, oxygen, nitrogen)
- List of nonmetal monographs
- Metallization pressure
- Nonmetal (astrophysics)
- Period 1 elements (hydrogen & helium)
- Properties of nonmetals (and metalloids) by group
Notes
Citations
Bibliography
External links
- Media related to Nonmetals at Wikimedia Commons