p-Xylene (data page)
In-game article clicks load inline without leaving the challenge.
This page provides supplementary chemical data on p-xylene.
Material Safety Data Sheet
The handling of this chemical may incur notable safety precautions. It is highly recommend that you seek the Material Safety Datasheet (MSDS) for this chemical from a reliable source and follow its directions.
Structure and properties
| Structure and properties | |
|---|---|
| Index of refraction, nD | 1.4958 at 20 °C |
| Dielectric constant, εr | 2.2 ε0 at 20 °C |
| Surface tension | 29.92 dyn/cm at 5 °C 28.27 dyn/cm at 20 °C 24.2 dyn/cm at 60 °C |
| Viscosity | 0.7385 mPa·s at 10 °C 0.6475 mPa·s at 20 °C 0.5134 mPa·s at 40 °C 0.3519 mPa·s at 80 °C 0.2424 mPa·s at 130 °C |
| Solubility | 0.160 g/L at 0 °C 0.181 g/L at 25 °C 0.22 g/L at 40 °C |
Thermodynamic properties
| Phase behavior | |
|---|---|
| Triple point | 286.3 K (13.15 °C), ? Pa |
| Critical point | 617 K (344 °C), 3500 kPa |
| Std enthalpy change of fusion, ΔfusHo | 17.1 kJ/mol |
| Std entropy change of fusion, ΔfusSo | 59.8 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapHo | 35.7 kJ/mol at 138 °C |
| Std entropy change of vaporization, ΔvapSo | ? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfHosolid | ? kJ/mol |
| Standard molar entropy, Sosolid | ? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfHoliquid | −24.4 kJ/mol |
| Standard molar entropy, Soliquid | 247 J/(mol K) |
| Enthalpy of combustion, ΔcHo | −4552 kJ/mol |
| Heat capacity, cp | 181.7 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfHogas | 1.796000E+04 kJ/kg-mol at 25 °C |
| Standard molar entropy, Sogas | ? J/(mol K) |
| Heat capacity, cp | 163.2 J/(mol K) at 120 °C |
| van der Waals' constants | a = 3134 L2 kPa/mol2 b = 0.1809 liter per mole |
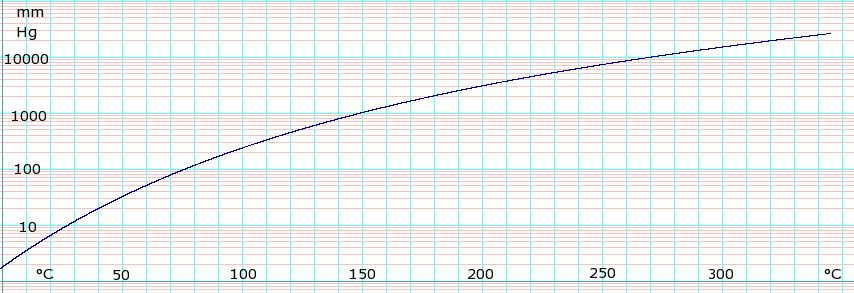
Vapor pressure of liquid
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 |
| T in °C | −8.1 | 27.3 | 54.4 | 75.9 | 115.9 | 138.3 |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed.
Distillation data
| BP Temp. °C | % by mole p-xylene | |||||
|---|---|---|---|---|---|---|
| Vapor-liquid Equilibrium for p-Xylene/o-Xylene P = 26.66 kPa BP Temp. °C % by mole p-xylene liquid vapor 100.1 0.0 0.0 99.8 4.9 5.8 99.5 9.9 11.8 99.0 17.4 20.7 98.5 25.1 29.3 98.1 32.6 37.1 97.6 40.6 44.9 97.2 48.6 53.8 96.7 56.9 61.8 96.2 64.9 69.6 95.8 73.1 77.3 95.4 81.4 84.5 94.9 90.7 91.6 94.6 95.4 96.0 94.4 100.0 100.0 | Vapor-liquid Equilibrium for p-Xylene/m-Xylene P = 26.66 kPa BP Temp. °C % by mole p-xylene liquid vapor 95.30 0.0 0.0 95.20 8.3 8.5 95.15 15.5 15.8 95.05 24.3 24.9 95.00 33.0 33.6 94.90 40.9 41.7 94.85 49.0 49.7 94.75 57.1 57.9 94.70 65.3 66.1 94.60 73.8 74.3 94.55 81.7 82.2 94.50 89.5 89.9 94.45 94.8 94.8 94.40 100.0 100.0 | Vapor-liquid Equilibrium for p-Xylene/carbon tetrachloride P = 760 mm Hg BP Temp. °C % by mole carbon tetrachloride liquid vapor 136.4 1.7 5.9 131.1 6.3 21.6 126.2 11.1 34.9 121.8 15.7 45.3 117.6 20.3 53.9 113.5 25.2 61.6 109.8 30.1 67.9 106.2 35.1 73.1 102.7 40.5 77.9 102.2 41.3 78.7 98.1 48.5 83.6 94.4 55.8 87.6 91.1 62.9 90.8 88.0 70.1 93.4 85.2 77.2 95.4 82.6 84.0 96.9 80.0 91.1 98.4 77.6 97.7 99.7 | Vapor-liquid Equilibrium for p-Xylene/Butanone P = 760 mm Hg BP Temp. °C % by mole butanone liquid vapor 129.45 4.8 20.8 125.42 7.9 31.7 123.25 10.2 38.1 116.80 16.1 51.1 112.44 21.0 59.4 105.55 30.5 71.0 101.15 38.7 75.5 95.30 50.6 85.2 93.90 54.6 87.2 89.45 67.0 91.7 86.55 77.0 94.7 85.02 81.5 95.9 84.58 83.1 96.2 80.65 97.0 994 Vapor-liquid Equilibrium for p-Xylene/Aniline P = 745 mm Hg BP Temp. °C % by mole p-xylene liquid vapor 171 7.5 37.0 165 14.0 50.0 156 26.5 67.5 148 48.5 80.0 142 72.5 89.0 140 83.0 92.0 | |||
| Vapor-liquid Equilibrium for p-Xylene/o-Xylene P = 26.66 kPa | ||||||
| liquid | vapor | |||||
| 100.1 | 0.0 | 0.0 | ||||
| 99.8 | 4.9 | 5.8 | ||||
| 99.5 | 9.9 | 11.8 | ||||
| 99.0 | 17.4 | 20.7 | ||||
| 98.5 | 25.1 | 29.3 | ||||
| 98.1 | 32.6 | 37.1 | ||||
| 97.6 | 40.6 | 44.9 | ||||
| 97.2 | 48.6 | 53.8 | ||||
| 96.7 | 56.9 | 61.8 | ||||
| 96.2 | 64.9 | 69.6 | ||||
| 95.8 | 73.1 | 77.3 | ||||
| 95.4 | 81.4 | 84.5 | ||||
| 94.9 | 90.7 | 91.6 | ||||
| 94.6 | 95.4 | 96.0 | ||||
| 94.4 | 100.0 | 100.0 | ||||
| Vapor-liquid Equilibrium for p-Xylene/m-Xylene P = 26.66 kPa | ||||||
| BP Temp. °C | % by mole p-xylene | |||||
| liquid | vapor | |||||
| 95.30 | 0.0 | 0.0 | ||||
| 95.20 | 8.3 | 8.5 | ||||
| 95.15 | 15.5 | 15.8 | ||||
| 95.05 | 24.3 | 24.9 | ||||
| 95.00 | 33.0 | 33.6 | ||||
| 94.90 | 40.9 | 41.7 | ||||
| 94.85 | 49.0 | 49.7 | ||||
| 94.75 | 57.1 | 57.9 | ||||
| 94.70 | 65.3 | 66.1 | ||||
| 94.60 | 73.8 | 74.3 | ||||
| 94.55 | 81.7 | 82.2 | ||||
| 94.50 | 89.5 | 89.9 | ||||
| 94.45 | 94.8 | 94.8 | ||||
| 94.40 | 100.0 | 100.0 | ||||
| Vapor-liquid Equilibrium for p-Xylene/carbon tetrachloride P = 760 mm Hg | ||||||
| BP Temp. °C | % by mole carbon tetrachloride | |||||
| liquid | vapor | |||||
| 136.4 | 1.7 | 5.9 | ||||
| 131.1 | 6.3 | 21.6 | ||||
| 126.2 | 11.1 | 34.9 | ||||
| 121.8 | 15.7 | 45.3 | ||||
| 117.6 | 20.3 | 53.9 | ||||
| 113.5 | 25.2 | 61.6 | ||||
| 109.8 | 30.1 | 67.9 | ||||
| 106.2 | 35.1 | 73.1 | ||||
| 102.7 | 40.5 | 77.9 | ||||
| 102.2 | 41.3 | 78.7 | ||||
| 98.1 | 48.5 | 83.6 | ||||
| 94.4 | 55.8 | 87.6 | ||||
| 91.1 | 62.9 | 90.8 | ||||
| 88.0 | 70.1 | 93.4 | ||||
| 85.2 | 77.2 | 95.4 | ||||
| 82.6 | 84.0 | 96.9 | ||||
| 80.0 | 91.1 | 98.4 | ||||
| 77.6 | 97.7 | 99.7 | ||||
| Vapor-liquid Equilibrium for p-Xylene/Butanone P = 760 mm Hg BP Temp. °C % by mole butanone liquid vapor 129.45 4.8 20.8 125.42 7.9 31.7 123.25 10.2 38.1 116.80 16.1 51.1 112.44 21.0 59.4 105.55 30.5 71.0 101.15 38.7 75.5 95.30 50.6 85.2 93.90 54.6 87.2 89.45 67.0 91.7 86.55 77.0 94.7 85.02 81.5 95.9 84.58 83.1 96.2 80.65 97.0 994 | ||||||
| Vapor-liquid Equilibrium for p-Xylene/Butanone P = 760 mm Hg | ||||||
| BP Temp. °C | % by mole butanone | |||||
| liquid | vapor | |||||
| 129.45 | 4.8 | 20.8 | ||||
| 125.42 | 7.9 | 31.7 | ||||
| 123.25 | 10.2 | 38.1 | ||||
| 116.80 | 16.1 | 51.1 | ||||
| 112.44 | 21.0 | 59.4 | ||||
| 105.55 | 30.5 | 71.0 | ||||
| 101.15 | 38.7 | 75.5 | ||||
| 95.30 | 50.6 | 85.2 | ||||
| 93.90 | 54.6 | 87.2 | ||||
| 89.45 | 67.0 | 91.7 | ||||
| 86.55 | 77.0 | 94.7 | ||||
| 85.02 | 81.5 | 95.9 | ||||
| 84.58 | 83.1 | 96.2 | ||||
| 80.65 | 97.0 | 994 | ||||
| Vapor-liquid Equilibrium for p-Xylene/Aniline P = 745 mm Hg BP Temp. °C % by mole p-xylene liquid vapor 171 7.5 37.0 165 14.0 50.0 156 26.5 67.5 148 48.5 80.0 142 72.5 89.0 140 83.0 92.0 | ||||||
| Vapor-liquid Equilibrium for p-Xylene/Aniline P = 745 mm Hg | ||||||
| BP Temp. °C | % by mole p-xylene | |||||
| liquid | vapor | |||||
| 171 | 7.5 | 37.0 | ||||
| 165 | 14.0 | 50.0 | ||||
| 156 | 26.5 | 67.5 | ||||
| 148 | 48.5 | 80.0 | ||||
| 142 | 72.5 | 89.0 | ||||
| 140 | 83.0 | 92.0 |
Spectral data
| UV-Vis | |
|---|---|
| Spectrum | |
| λmax | ? nm |
| Extinction coefficient, ε | ? |
| IR | |
| Spectrum | |
| Major absorption bands | 793.94 cm−1 |
| NMR | |
| Spectrum | [permanent dead link] |
| Proton NMR | 2.296, 7.046 |
| Carbon-13 NMR | 134.66, 128.97, 20.90 |
| MS | |
| Spectrum | |
| Masses of main fragments | 106, 91, 77 |
- Except where noted otherwise, data relate to Standard temperature and pressure.
- Reliability of data general note.
Notes
Bibliography
- . Archived from on 13 January 2004.
- . Archived from on 5 May 2006.