Prime editing
In-game article clicks load inline without leaving the challenge.
Prime editing is a 'search-and-replace' genome editing technology in molecular biology by which the genome of living organisms may be modified. The technology directly writes new genetic information into a targeted DNA site. It uses a fusion protein, consisting of a catalytically impaired Cas9 endonuclease fused to an engineered reverse transcriptase enzyme, and a prime editing guide RNA (pegRNA), capable of identifying the target site and providing the new genetic information to replace the target DNA nucleotides. It mediates targeted insertions, deletions, and base-to-base conversions without the need for double strand breaks (DSBs) or donor DNA templates.
The technology has received mainstream press attention due to its potential uses in medical genetics. It utilizes methodologies similar to precursor genome editing technologies, including CRISPR/Cas9 and base editors. Prime editing has been used on some animal models of genetic disease and plants. In 2024, PM359, a gene therapy developed by Prime Medicine, became the first prime editor to enter clinical trials for human use. Prime Medicine reported in December 2025 that two chronic granulomatous disease patients treated with PM359 had been "effectively cured" of the disease.
Components
Cas9 prime editors
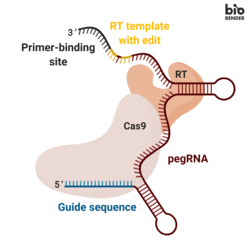
Prime editing involves three major components:
- A prime editing guide RNA (pegRNA), capable of (i) identifying the target nucleotide sequence to be edited, and (ii) encoding new genetic information that replaces the targeted sequence. The pegRNA consists of an extended single guide RNA (sgRNA) containing a primer binding site (PBS) and a reverse transcriptase (RT) template sequence. During genome editing, the primer binding site allows the 3’ end of the nicked DNA strand to hybridize to the pegRNA, while the RT template serves as a template for the synthesis of edited genetic information.
- A fusion protein consisting of a Cas9 H840A nickase fused to a Moloney Murine Leukemia Virus (M-MLV) reverse transcriptase. Cas9 H840A nickase: the Cas9 enzyme contains two nuclease domains that can cleave DNA sequences, a RuvC domain that cleaves the non-target strand and a HNH domain that cleaves the target strand. The introduction of a H840A substitution in Cas9, through which the 840th amino acid histidine is replaced by an alanine, inactivates the HNH domain. With only the RuvC functioning domain, the catalytically impaired Cas9 introduces a single strand nick, hence the name nickase. M-MLV reverse transcriptase: an enzyme that synthesizes DNA from a single-stranded RNA template.
- A single guide RNA (sgRNA) that directs the Cas9 H840A nickase portion of the fusion protein to nick the non-edited DNA strand.
Accessory components not present in all editors:
- RNA-binding protein
Cas12 prime editors
Involves more components than a Cas9 prime editor
Editing mechanism

Original (used by PE1 and 2)
Genomic editing takes place by transfecting cells with the pegRNA and the fusion protein. Transfection is often accomplished by introducing vectors into a cell. Once internalized, the fusion protein nicks the target DNA sequence, exposing a 3’-hydroxyl group that can be used to initiate (prime) the reverse transcription of the RT template portion of the pegRNA. This results in a branched intermediate that contains two DNA flaps: a 3’ flap that contains the newly synthesized (edited) sequence, and a 5’ flap that contains the dispensable, unedited DNA sequence. The 5’ flap is then cleaved by structure-specific endonucleases or 5’ exonucleases. This process allows 3’ flap ligation, and creates a heteroduplex DNA composed of one edited strand and one unedited strand. The reannealed double stranded DNA contains nucleotide mismatches at the location where editing took place. In order to correct the mismatches, the cells exploit the intrinsic mismatch repair (MMR) mechanism, with two possible outcomes: (i) the information in the edited strand is copied into the complementary strand, permanently installing the edit; (ii) the original nucleotides are re-incorporated into the edited strand, excluding the edit.

PE3 strategy
Despite its increased efficacy, the edit installed by PE2 might still be removed by mismatch repair of the edited strand. To counter this, the Prime Editor 3 strategy comes with an extra component borrowed from base editing. In addition to the editor itself, PE3 uses a standard Cas9 nickase to exploit DNA repair systems by targeting the opposite non-edited strand. Following the nick on the non-edited strand, DNA repair will use the edited strand as a template, consequentially improving the odds of success.
However, there are drawbacks to this system as the unaltered strand can be nicked prematurely and lead to additional undesired indels.
Paired prime editing
HOPE strategy: use two complimentary guide RNA pairs to increase chances of edit inclusion (just like Rin and Len guys!)[more detail needed]
PE4 and 5 strategies
Prime editor 4 utilizes the same machinery as PE2, but also includes a plasmid that encodes for dominant negative MMR protein MLH1. Dominant negative MLH1 is able to essentially knock out endogenous MLH1 by inhibition, thereby reducing cellular MMR response and increasing prime editing efficiency.[more detail needed]
Prime editor 5 utilizes the same machinery as PE3, but also includes a plasmid that encodes for dominant negative MLH1. Like PE4, this allows for a knockdown of endogenous MMR response, increasing the efficiency of prime editing.[more detail needed]
Twin prime editing
The "twin prime editing" (twinPE) mechanism reported in 2021 allows editing large sequences of DNA – sequences as large as genes – which addresses the method's key drawback. It uses a prime editor protein and two prime editing guide RNAs.[more detail needed]
PASTE
Drag and drop using an Integrase[more detail needed]
PASSIGE
Performs way better than PASTE[more detail needed]
Structural improvements and modifications
Prime editing was developed in the lab of David R. Liu at the Broad Institute and disclosed in Anzalone et al. (2019). Since then prime editing and the research that produced it have received widespread scientific acclaim, being called "revolutionary" and an important part of the future of editing. During the development of this technology, several modifications were done to the components, in order to increase its effectiveness.
Prime editor 1 (PE1)
Prime editor 1 was a prototype designed to have the minimum components necessary. It consisted of a wild-type Moloney murine leukemia virus (M-MLV) reverse transcriptase attached to Cas9 H840A nickase by a flexible protein linker. Originally, the linker was too short and the reverse transcriptase was unable to do its job. However, extending it to 8-15 bases led to detectable editing. During development, the linker was attached to either the C- or N-terminal of the Cas9 protein. Linkage the C terminal proved more efficient, so it was chosen as the final design. Detectable editing efficiencies were observed.
Prime editor 2 (PE2)
After the success of PE1, researchers chose to modify its reverse transcriptase. This was done to improve DNA synthesis and thus the editor's overall efficiency. They started by searching the literature for previously described mutations. These were found to improve the enzyme's thermostability, processivity, DNA to RNA substrate affinity, or prevent interference by RNaseH. A variety of mutations were combined into nineteen different variants and then tested against each other in human cells.
Researchers began by focusing on thermal stability. They found that a reverse transcriptase combining D200N, L603W, and T330P mutations improved editing efficiency by an average of 6.8-fold for insertions and transversions at five different sites. Following this, the mutant transcriptase was further combined with additional mutations. Adding T306K and W313F improved editing efficiency by an additional 1.3 to 3-fold for insertions or transversions at the same five sites. The resulting prime editor was deemed PE2.
Prime Editor 2 had a substantially higher efficiency than its predecessor when making insertion, deletion, or substitution mutations. Additionally, it was compatible with a shorter primer binding site (PBS) sequence due to its enhanced substrate affinity.
PAM flexible prime editors
study[more detail needed]
PE2*
better PE2 with enhanced nuclear localization sequences
Engineered pegRNA (epegRNA)
Prime editing efficiency can be increased with the use of engineered pegRNAs (epegRNAs). One common issue with traditional pegRNAs is degradation of the 3' end, leading to decreased PE efficiency. epegRNAs have a structured RNA motif added to their 3' end to prevent degradation.[more detail needed]
PEmax
PEmax was created to further enhance the improvements made in PE2. To do this, researchers tested twenty-one PE2 variants with different Cas9 mutations, reverse transcriptase codon usages, nuclear localization sequences (NLS), protein linker lengths, and protein linker compositions. Out of the twenty-one created, an editor with a human-optimized reverse transcriptase, Cas9 with the nucelase enhancing R221K and N394K mutations, 34-aa linker with a bipartite SV40 NLS, and an additional c-Myc NLS at the protein's C-terminal emerged most successful.
Afterwards, the resulting protein (dubbed PEmax) was pitted against other improved prime editors. All were tested on HeLa cells and performed various modifications at seven genomic sites. Notable competitors included PE2*, which has additional NLS sequences, and CMP–PE–V1, which contains high-mobility peptides. PEmax outperformed both at 6/7 sites, with the time it lost to PE2* being within the margin of error.
While improvements vary by cell line and modification, PEmax can be combined with various strategies (PE2max, PE3max, PE4max, PE5max) as well as epegRNA to dramatically enhance editing efficiency. However, note that combining PEmax with PE3 or PE5 leads to a slight increase in indels. This is likely due to the editor's increased nicking ability.
Nuclease prime editor (PEn)
Nuclease Prime Editor uses Cas9 nuclease instead of Cas9(H840A) nickase. Unlike prime editor 3 (PE3) that requires dual-nick at both DNA strands to induce efficient prime editing, Nuclease Prime Editor requires only a single pegRNA since the single-gRNA already creates double-strand break instead of single-strand nick.[more detail needed]
further research done?[more detail needed]
Upgraded nuclease prime editor (uPEn)
study[more detail needed]
Prime editor 6 (PE6)
Smaller than normal. There are multiple variants that can be used in different applications (a,b,c,d)[more detail needed]
Yeast-improved prime editor (PE_Y18)
study[more detail needed]
Prime editor 7 (PE7)
study[more detail needed]
Cas12a prime editors
source[more detail needed]
Applications
Cell cultures
Animal models
Efficient prime editing in mouse brain,liver and heart with dual AAVs[more detail needed]
zebrafish[more detail needed]
Agriculture
PE is among recently introduced technologies which allow the transfer of single-nucleotide polymorphisms (SNPs) from one individual crop plant to another. PE is precise enough to be used to recreate an arbitrary SNP in an arbitrary target, including deletions, insertions, and all 12 point mutations without also needing to perform a double-stranded break or carry a donating template.
Ultra-efficient tomato prime editor[more detail needed]
rice[more detail needed]
Gene therapy
Other
Prime editors may be used in gene drives. A prime editor may be incorporated into the Cleaver half of a Cleave and Rescue ClvR system. In this case it is not meant to perform a precise alteration but instead to merely disrupt.[more detail needed]
Methods of delivery
Base editors used for prime editing require delivery of both a protein and RNA molecule into living cells. Introducing exogenous gene editing technologies into living organisms is a significant challenge. One potential way to introduce a base editor into animals and plants is to package the base editor into a viral capsid. The target organism can then be transduced by the virus to synthesize the base editor in vivo. Common laboratory vectors of transduction such as lentivirus cause immune responses in humans, so proposed human therapies often centered around adeno-associated virus (AAV) because AAV infections are largely asymptomatic. Unfortunately, the effective packaging capacity of AAV vectors is small, approximately 4.4kb not including inverted terminal repeats. As a comparison, an SpCas9-reverse transcriptase fusion protein is 6.3kb, which does not even account for the lengthened guide RNA necessary for targeting and priming the site of interest. However, successful delivery in mice has been achieved by splitting the editor into two AAV vectors or by using an adenovirus, which has a larger packaging capacity.
Comparison to base editors and traditional CRISPR Cas9 genome editing
Although additional research is required to improve the efficiency of prime editing, the technology offers promising scientific improvements over other gene editing tools. The prime editing technology has the potential to correct the vast majority of pathogenic alleles that cause genetic diseases, as it can repair insertions, deletions, and nucleotide substitutions.
Advantages
The prime editing tool offers advantages over traditional gene editing technologies. CRISPR/Cas9 edits rely on double-strand breaks and non-homologous end joining (NHEJ) or homology-directed repair (HDR) to fix DNA breaks, while the prime editing system employs single-strand breaks and DNA mismatch repair. This is an important feature of this technology given that DNA repair mechanisms such as NHEJ and HDR, generate unwanted, random insertions or deletions (indels). These are byproducts that complicate the retrieval of cells carrying the correct edit. Prime editors do not frequently create these indel byproducts, suggesting that prime editors can be more precise than earlier tools.
The prime editing system introduces single-stranded DNA breaks, as with base editors, instead of the double-stranded DNA breaks observed in other editing tools, such as CRISPR/Cas9 editing. Collectively, base editing and prime editing offer complementary strengths and weaknesses for making targeted transition mutations. Base editors offer higher editing efficiency and fewer indel byproducts if the desired edit is a transition point mutation and a PAM sequence exists roughly 15 bases from the target site. However, because the prime editing technology does not require a precisely positioned PAM sequence to target a nucleotide sequence, it offers more flexibility and editing precision. Remarkably, prime editors allow all types of substitutions, both transitions and transversions, to be installed into the target sequence. Cytosine base editing and adenine BE can already perform precise base transitions but base transversions cannot be achieved with these base editors. Prime editing performs transversions with high efficiency. PE can insert up to 44bp, delete up to 80, or combinations thereof.
Because the prime system involves three separate DNA binding events (between (i) the guide sequence and the target DNA, (ii) the primer binding site and the target DNA, and (iii) the 3’ end of the nicked DNA strand and the pegRNA), it has been suggested to have fewer undesirable off-target effects than CRISPR/Cas9.
Limitations
There is considerable interest in applying gene-editing methods to the treatment of diseases with a genetic component. However, there are multiple challenges associated with this approach. An effective treatment would require editing of a large number of target cells, which in turn would require an effective method of delivery and a great level of tissue specificity.
As of 2019, prime editing looks promising for relatively small genetic alterations, but more research needs to be conducted to evaluate whether the technology is efficient in making larger alterations, such as targeted insertions and deletions. Larger genetic alterations would require a longer RT template, which could hinder the efficient delivery of pegRNA to target cells. Furthermore, a pegRNA containing a long RT template could become vulnerable to damage caused by cellular enzymes. Prime editing in plants suffers from low efficiency ranging from zero to a few percent and needs significant improvement.
Some of these limitations have been mitigated by recent improvements to the prime editors, including motifs that protect pegRNAs from degradation. Further research is needed before prime editing could be used to correct pathogenic alleles in humans. Research has also shown that inhibition of certain MMR proteins, including MLH1 can improve prime editing efficiency.